The synthesis of Tirzepatide
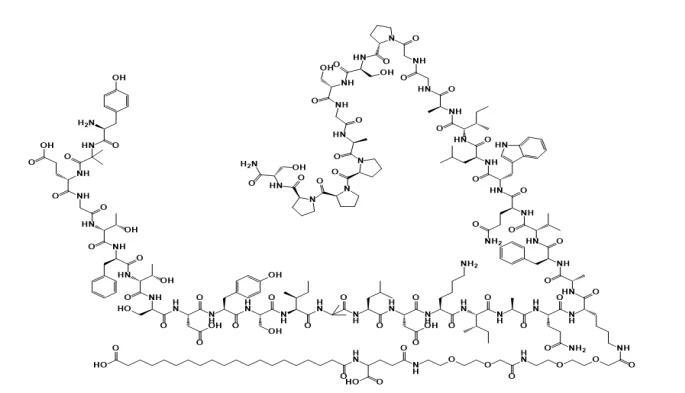
The synthesis of Tirzepatide mainly adopts the strategy of solid-phase fragment condensation and solid-liquid binding, which focuses on solving the static hindrance effect of unnatural amino acids (such as Aib) in its 39 amino acid long chains, complex modifications of side chains (such as C20 diacid-PEG-γ-Glu-double AEEA-Lys²⁰), and racemic impurity control. The following are specific synthesis methods and technical points:
Solid phase fragment condensation strategy (including Aib fragment presynthesis)
In order to solve the problems of low coupling efficiency and racemic impurities caused by steric hindrance of Aib (α-aminoisobutyric acid), presynthesized short peptide fragments containing Aib were used to replace single amino acids for stepwise coupling:
- Key Fragment Design:
• N-terminal fragments: Use 2-6 peptide fragments (e.g., Tyr-Aib, Tyr-Aib-Glu-Gly, or Tyr-Aib-Glu-Gly-Thr-Phe) to access amino acids 1-5.
• C-terminal fragments: Ile-Aib, Ile-Aib-Leu-Asp, and other fragments are used to access the 12th-15th amino acids. - Synthesis Process:
• 2-chlorotriphenyl (2-CTC) resin was used as a carrier to gradually conjugate the fragments through the Fmoc/tBu protection strategy.
•Example:
Synthesis of Boc-Tyr(tBu)-Aib-OH: Fmoc-Aib-2-CTC resin is deprotected and conjugated to Boc-Tyr(tBu)-OH to obtain dipeptides.
Synthetic Fmoc-Ile-Aib-OH: A similar method is used to couple Fmoc-Ile-OH with Aib resin. - Advantages:
Reduce racemic impurities directly conjugated to Aib (e.g., Thr⁵, Thr⁷ deletion peptides), and increase the purity of crude peptides to more than 70%.
Solid-liquid combination two-fragment method (mass production core)
In order to solve the problem of long-chain synthesis and purification, 39 peptides were split into fragments 1-18 and 19-39, and the liquid phase was coupled after synthesis respectively:
- Fragment 19-39 Synthesis:
Solid-phase synthesis from the C-terminal (Ser³⁹) using Sieber resin as a carrier.
Key step: Introduction of side-chain modified monomer Fmoc-Gln(Trt)-Lys(N-ε-AEEA-AEEA-γGlu-C20-diacid)-OH (requires copper ion catalytic coupling). - Synthesis of Fragments 1-18:
Using CTC resin as the carrier, the N-terminal starts with Boc-Tyr(tBu)-Aib-OH and gradually connects to Fmoc-Glu(OtBu)-Gly-OH. - Liquid phase coupling and deprotection:
• Fragments 1-18 (carboxyl terminus) and fragments 19-39 (amino terminus) are conjugated under the action of a condensing agent (e.g., HATU/DIPEA).
• The protective peptide is removed by trifluoroacetic acid (TFA) cleavage system (e.g., TFA:EDT:TIS=90:5:5). - Advantages:
• The crude peptide yield was 46%, and the total yield after purification was significantly higher than that of all-solid-phase synthesis.
Pseudoproline peptide strategy (reduce racema of difficult-to-synthesize sites)
For racemic-prone sites (e.g., Ser/Thr dense regions), a pseudoproline peptide (PsiPro) structure is used:
• Key dipeptide introduction:
• 7th-8th place: Fmoc-Thr(tBu)-Ser(ΨMe, MePro)-OH
• 10th-11th place: Fmoc-Tyr(tBu)-Ser(ΨMe, MePro)-OH
• 32nd-33rd position: Fmoc-Ser(tBu)-Ser(ΨMe, MePro)-OH
•Effect:
• Inhibits β elimination of Ser/Thr, reduces racemic impurities, and has a purity of > 95% for crude peptide HPLC.
Cracking and purification process
- Cracking System Optimization:
• Standard lysate: TFA:EDT:TIS=90:5:5 (with scavenger to reduce side effects).
• Complex side chain cleavage: TFA: Phenol: H₂O: Anine ether: EDT=82.5:5:5:5:5:2.5 to prevent macromolecular side chain modification breakage. - Purification Techniques:
• Nanofiltration: Separates low molecular weight impurities and avoids the gelation problems of traditional precipitation methods.
• Chromatographic purification: Reversed-phase HPLC (C18 column) gradient elution with ≥99% purity after lyophilization.
Mass production strategy (hybrid SPPS/LPPS)
- Four-fragment continuous flow synthesis:
• Fragment division: 1-14, 15-21, 22-29, 30-39 (shorten the synthesis cycle of individual fragments).
• Flow reactor coupling: Fragments are continuously condensed in a microchannel reactor to monitor the coupling efficiency in real time. - Industrialization Advantages:
• Solvent consumption is reduced by 50% and synthesis cycles are shortened by 30%, making it suitable for kilogram-level production.
For any enquiries about P29/diapeptides/side chain/semaglutide api/tirzepatide api, plz feel free to contact me.
Tel: +8618575536586
Email: haoran.tse@gmail.com
评论已关闭